
Īs a final example: nitrous oxide is 63.3% nitrogen and 36.7% oxygen, nitric oxide is 44.05% nitrogen and 55.95% oxygen, and nitrogen dioxide is 29.5% nitrogen and 70.5% oxygen. In these respective oxides, for every two atoms of iron, there are two or three atoms of oxygen ( Fe 2O 2 and Fe 2O 3). Adjusting these figures, in the black oxide there is about 28 g of oxygen for every 100 g of iron, and in the red oxide there is about 42 g of oxygen for every 100 g of iron. There is one type of iron oxide that is a black powder which is 78.1% iron and 21.9% oxygen and there is another iron oxide that is a red powder which is 70.4% iron and 29.6% oxygen. 
In these oxides, for every tin atom there are one or two oxygen atoms respectively ( SnO and SnO 2). Adjusting these figures, in the black oxide there is about 13.5 g of oxygen for every 100 g of tin, and in the white oxide there is about 27 g of oxygen for every 100 g of tin. This pattern suggested to Dalton that each chemical element combines with other elements by some basic and consistent unit of mass, and he decided to call these units "atoms".įor example, there are two types of tin oxide: one is a black powder that is 88.1% tin and 11.9% oxygen, and the other is a white powder that is 78.7% tin and 21.3% oxygen. He noticed that in chemical compounds which contain a particular chemical element, the content of that element in these compounds will differ by ratios of small whole numbers. In the early 1800s, the English chemist John Dalton compiled experimental data gathered by himself and other scientists and discovered a pattern now known as the " law of multiple proportions". Chemistry is the discipline that studies these changes.Ītoms and molecules as depicted in John Dalton's A New System of Chemical Philosophy vol. The ability of atoms to attach and detach is responsible for most of the physical changes observed in nature. For example, New York City's Statue of Liberty was originally made of pure copper, but over the years, the surface combined with oxygen, carbon and sulfur atoms to make a green patina on the copper. For example, a copper atom with 34 neutrons is copper-63 (29+34), and with 36 neutrons is copper-65 natural copper is about 70% Cu-63 and the rest is Cu-65.Ītoms can attach to one or more other atoms by chemical bonds to form chemical compounds such as molecules or crystals. The number of neutrons defines the isotope of the element. For example, any atom that contains 29 protons is copper. The number of protons in the nucleus is the atomic number and it defines to which chemical element the atom belongs. In this case, the nucleus splits and leaves behind different elements. 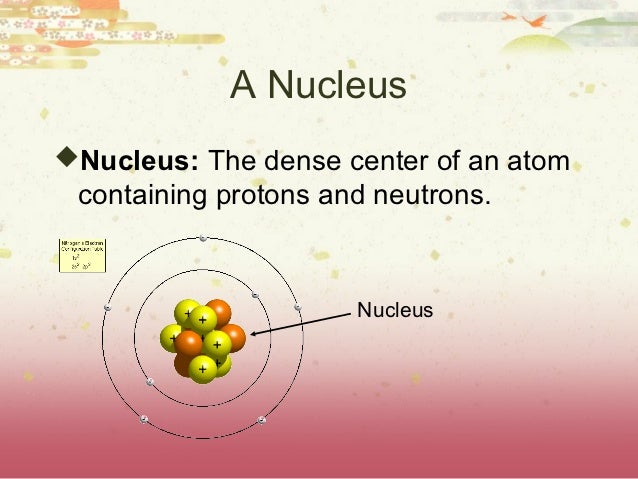
Under certain circumstances, the repelling electromagnetic force becomes stronger than the nuclear force. This force is usually stronger than the electromagnetic force that repels the positively charged protons from one another. The protons and neutrons in the nucleus are attracted to each other by the nuclear force. The electrons of an atom are attracted to the protons in an atomic nucleus by the electromagnetic force. If an atom has more or fewer electrons than protons, then it has an overall negative or positive charge, respectively – such atoms are called ions. If the number of protons and electrons are equal, then the atom is electrically neutral. The protons have a positive electric charge, the electrons have a negative electric charge, and the neutrons have no electric charge. More than 99.94% of an atom's mass is in the nucleus. Only the most common variety of hydrogen has no neutrons. The nucleus is made of one or more protons and a number of neutrons. They are so small that accurately predicting their behavior using classical physics-as if they were tennis balls, for example-is not possible due to quantum effects.Įvery atom is composed of a nucleus and one or more electrons bound to the nucleus. Atoms are extremely small, typically around 100 picometers across. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Smallest recognized division of a chemical elementĮlectrons and a compact nucleus of protons and neutronsĪn atom is the smallest unit of ordinary matter that forms a chemical element. The black bar is one angstrom ( 10 −10 m or 100 pm). The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. An illustration of the helium atom, depicting the nucleus (pink) and the electron cloud distribution (black).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
